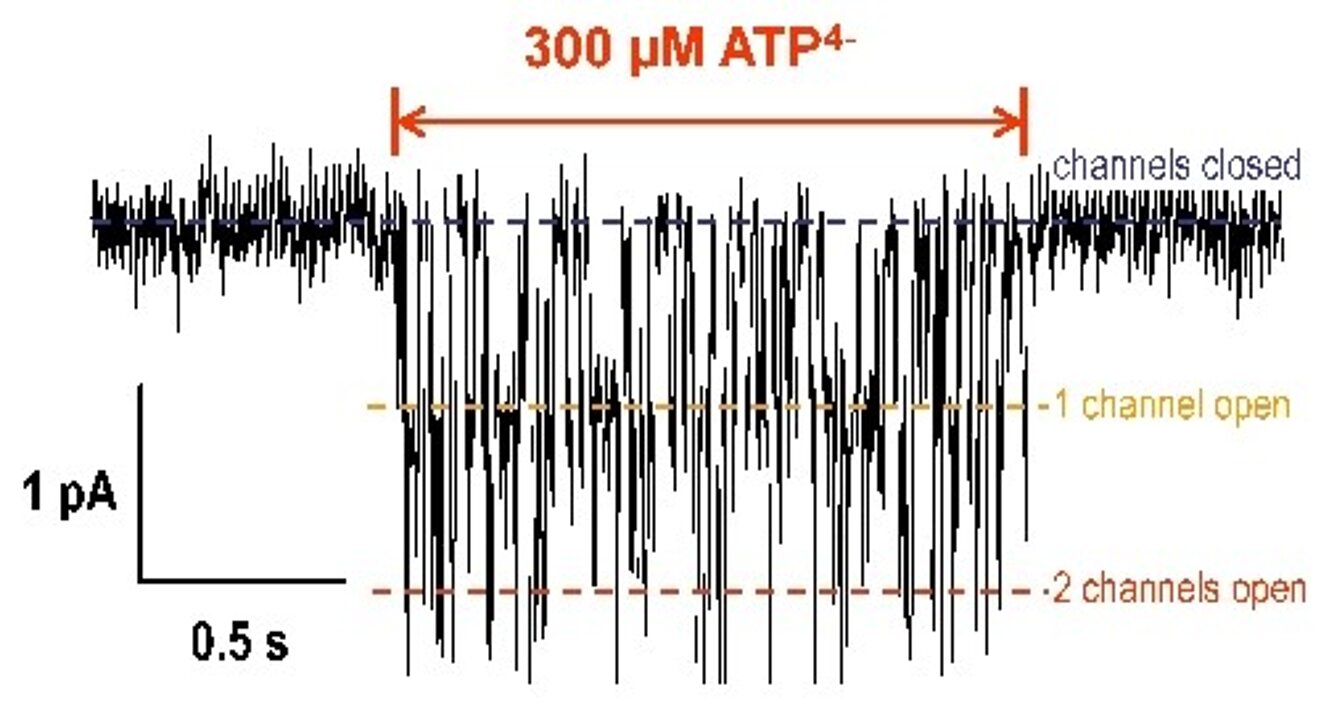
Structure and function of purinergic P2X receptors
Purinergic P2X receptors are cationic channels in the cell membrane which are opened by extracellular ATP. They are widely expressed in different tissues like skeletal muscle, endothelial cells, neurons and leukocytes. ATP is released from nerve endings into the extracellular space as a neurotransmitter and from several other cells during pathophysiological conditions like hypoxia, thrombosis and inflammation. We investigate the function of P2X receptors in lymphocytes and macrophages by means of the whole-cell and single-channel voltage clamp technique and by measurement of P2X-dependent changes in the membrane potential and the intracellular Ca2+ and Na+-concentration in single cells by voltage clamp fluorimetry and in cell populations using flow cytometry (FACS). In collaboration with molecular biologists (Prof. Günther Schmalzing, RWTH Aachen) mutated P2X receptor constructs are expressed in Xenopus oocytes and mammalian cells. Structure-function relationships of the receptors and the interaction of P2X receptors with other ion channels (pannexin, anion channels,trp channels) are investigated by the measurement of whole cell and single channel P2X-dependent ionic currents and fluorescence measurements.
Supported by the DFG and the Roux programme of the Medical Faculty of the Martin Luther University.
Mechanism of sphingosine phosphate-induced ATP secretion
Macrophyges are versatile players coordinating inflammatory and regenerative processes under pathological conditions in which sphingosine-1-phosphate (S1P)-mediated migration is essential. We investigated the involved signaling cascade by means of voltage clamp, measurement of ATP secretion, and wound healing assay.
M.sc. Malte Berthold (Ph.D. student)
Doctoral candidates:
Mira Trang
Eike Schön
Danyal Zahiri
Markus Schneider
Hannah Dentler
Ida Schiller
Thomas Kendzierski
- Modulation of Calcium channel current in guinea-pig single ventricular heart cells by the dihydropyridine Bay k 8644
F. Markwardt, B. Nilius
J. Physiol. (Lond.) 399 (1988), 559-575
PubMed - Thrombin stimulates Ca-channel currents in isolated frog ventricular cells
F. Markwardt, R. Albitz, T. Franke, B. Nilius
Pflügers Arch. 412 (1988), 668-670
PubMed - Effects of thrombin on single calcium channels in frog ventricular cells
F. Markwardt, T. Franke, R. Albitz, B. Nilius
Pflügers Arch. 415 (1990), 547-553
PubMed - Gating of maxi K+ channels studied by Ca2+ concentration jumps in excised inside-out multi-channel patches (myocytes from guinea-pig urinary bladder)
F. Markwardt, G. Isenberg
J. Gen. Physiol. 99 (1992), 841-862
PubMed - Nonselective cationic currents elicited by extracellular ATP in human B-lymphocytes
F. Bretschneider, M. Klapperstück, M. Löhn, F. Markwardt
Pflügers Arch. 429 (1995), 691-698
PubMed - Purinoceptor-operated cationic channels in human B lymphocytes
F. Markwardt, M. Löhn, T. Böhm, M. Klapperstück
J. Physiol. Lond. 498 (1997), 143-151
PubMed - Acetylcholine-induced K+-currents in smooth muscle cells of intact rat small arteries
T. Weidelt, W. Boldt, F. Markwardt
J. Physiol. Lond. 500 (1997), 617-630
PubMed - Antagonism by the suramin analogue NF279 on human P2X1 and P2X7 receptors
M. Klapperstück, C. Büttner, P. Nickel, G. Schmalzing, G. Lambrecht, F. Markwardt
Eur. J. Pharmacol. 387 (2000), 245-252
PubMed - Block by extracellular Mg2+ of single human purinergic P2X4 receptor channels expressed in human embryonic kidney cells
Y.A. Negulyaev, F. Markwardt
Neuroscience Lett. 279 (2000), 165-168
PubMed - Characteristics of P2X7 receptors from human B lymphocytes expressed in Xenopus oocytes
M. Klapperstück, C. Büttner, T. Böhm, G. Schmalzing, F. Markwardt
Biochim. Biophys. Acta 1467 (2000), 444-456
PubMed - Sodium block and depolarisation diminish P2Z-dependent Ca2+ entry in human B lymphocytes
M. Löhn, M. Klapperstück, D. Riemann, F. Markwardt
Cell Calcium 29 (2001), 395-408
PubMed - Functional evidence of distinct ATP activation sites at the human P2X7 receptor
M. Klapperstück, C. Büttner, G. Schmalzing, F. Markwardt
J. Physiol. Lond. 534 (2001), 25-35
PubMed - Desynchronizing effect of the endothelium on intracellular Ca2+ concentration dynamics in vascular smooth muscle cells of rat mesenteric arteries
M. Sell, W. Boldt, F. Markwardt
Cell Calcium 32 (2002), 105-120
PubMed - The Glu496Ala polymorphism of the human P2X7 receptor does not affect its electrophysiological phenotype
W. Boldt, M. Klapperstück, C. Büttner, S. Sadtler, G. Schmalzing, F. Markwardt
Am. J. Physiol. Cell Physiol. 284 (2003), C749-C756
PubMed - NF449, a novel picomolar potency antagonist at human P2X1 receptors
M. Hülsmann, P. Nickel, M. Kassack, G. Schmalzing, G. Lambrecht, F. Markwardt
Eur. J. Pharmacol. 470 (2003), 1-7
PubMed - Kinetics of P2X7 receptor-operated single channels currents
T. Riedel, I. Lozinsky, G. Schmalzing, F. Markwardt
Biophys. J. 92 (2007), 2377-2391
PubMed - Influence of extracellular monovalent cations on pore and gating properties of P2X7 receptor-operated single channels currents
T. Riedel, G. Schmalzing, F. Markwardt
Biophys. J. 93 (2007), 846-858
PubMed - The P2X7 carboxyl tail is a regulatory module of P2X7 receptor channel activity
D. Becker, R. Woltersdorf, W. Boldt, S. Schmitz, U. Braam, G. Schmalzing, F. Markwardt
J. Biol. Chem. 283 (2008), 25725-25734
PubMed - Sphingosine-1-phosphate receptors stimulate macrophage plasma membrane actin assembly via ADP release, ATP synthesis and P2X7 receptor activation
M.P. Kuhnel, M. Reiss, P. Anand, I. Treede, D. Holzer, E. Hoffmann, M. Klapperstueck, T. Steinberg, F. Markwardt, G. Griffiths
J. Cell Sci. (2009), 122, 505-512
PubMed - Effects of protons on macroscopic and single-channel currents mediated by the human P2X7 receptor
B. Flittiger, M. Klapperstück, G. Schmalzing, F. Markwardt
Biochim. Biophys. Acta Biomembranes 1798 (2010), 947-957
PubMed - Trophic activity of a naturally occurring truncated isoform of the P2X7 receptor
E. Adinolfi, M. Cirillo, R. Woltersdorf, S. Falzoni, P. Chiozzi, P. Pellegatti, M.G. Callegari, D. Sandonà, F. Markwardt, G. Schmalzing, F. Di Virgilio
FASEB J. 24 (2010), 3393-3404
PubMed - TMEM16A(a)/anoctamin-1 shares a homodimeric architecture with ClC chloride channels
G. Fallah, T. Römer, U. Braam, S. Detro-Dassen, F. Markwardt, G. Schmalzing
Mol. Cell. Proteomics 10 (2011), M110.004697
PubMed - The effect of anions on the human P2X7 receptor
C. Kubick, G. Schmalzing, F. Markwardt
Biochim. Biophys. Acta Biomembranes 1808 (2011), 2913-22
PubMed - Salt bridge switching from Arg290/Glu167 to Arg290/ATP promotes the closed-to-open transition of the P2X2 receptor
R. Hausmann, J. Gunther, A. Kless, D. Kuhlmann, M.U. Kassack, G. Bahrenberg, F. Markwardt, G. Schmalzing
Mol. Pharmacol. 83 (2013), 73-84
PubMed - Calibration procedures for the quantitative determination of membrane potential in human cells using anionic dyes
T. Klapperstück, D. Glanz, S. Hanitsch, M. Klapperstück, F. Markwardt, J. Wohlrab
Cytometry A 83 (2013), 612-626
PubMed - Activation of ATP secretion via volume-regulated anion channels by sphingosine-1-phosphate in RAW macrophages
P. Burow, M. Klapperstück, F. Markwardt
Pflügers Arch. (2015), 467, 1215-1226
PubMed - Homodimeric anoctamin-1, but not homodimeric anoctamin-6, is activated by calcium increases mediated by the P2Y1 and P2X7 receptors
M. Stolz, M. Klapperstück, T. Kendzierski, S. Detro-Dassen, A. Panning, G. Schmalzing, F. Markwardt
Pflügers Arch. 467 (2015), 2121-2140
PubMed - Inhibition of antigen receptor-dependent Ca2+ signals and NF-AT activation by P2X7 receptors in human B lymphocytes
A. Pippel, B. Beßler, M. Klapperstück, F. Markwardt
Cell Calcium 57 (2015), 275-289
PubMed - Localization of the gate and selectivity filter of the full-length P2X7 receptor
A. Pippel, M. Stolz, R. Woltersdorf, A. Kless, G. Schmalzing, F. Markwardt
Proc Natl Acad Sci U S A. 114 (2017), E2156-E2165
PubMed - Interaction of purinergic P2X4 and P2X7 receptor subunits
M. Schneider, K. Prudic, A. Pippel, M. Klapperstück, U. Braam, C.E. Müller, G. Schmalzing, F. Markwardt
Front. Pharmacol. 8 (2017), 860, doi: 10.3389/fphar.2017.00860
PubMed - The elusive P2X7 macropore
F. Di Virgilio, G. Schmalzing, F. Markwardt
Trends Cell Biol. 28 (2018), 392-404, 15,3
PubMed - Dissection of P2X4 and P2X7 receptor current components in BV-2 microglia
M. Trang, G. Schmalzing, C.E. Müller, F. Markwardt
Int.J.Mol.Sci. 21 (2020), 8489
https://doi.org/10.3390/ijms21228489 - Sphingosine-1-phosphate induces migration of microglial cells via activation of volume-sensitive anion channels, ATP secretion and activation of purinergic receptors
D. Zahiri, P. Burow, C. Großmann, C.E. Müller, M. Klapperstück, F. Markwardt
BBA - Molecular Cell Research (2020) 1868(2):118915
https://doi.org/10.1016/j.bbamcr.2020.118915 - Human P2X7 receptors - properties of single ATP-gated ion channels
F. Markwardt
Biochemical Pharmacology (2020) 114307
PubMed